
Advancing Radiotheranostics for Solid Cancer Treatment
A platform that identifies patients with Trop-2-expressing tumors using a diagnostic variant of an antibody, then treats them using the same antibody engineered into a targeted therapeutic.

There is a critical medical need for improved therapeutic options for patients with advanced metastatic solid tumors, which with few exceptions remain incurable. Although new systemic therapies have improved survival for some, major challenges persist because many contemporary targeted drugs including Antibody Drug Conjugates (ADCs) lack therapy-predictive biomarkers that pinpoint those with an optimal risk-benefit balance. As a result, therapeutic efficacy for unselected patients is still limited, and the risk for unnecessary toxicity is substantial.
PROBLEM
SOLUTION
Established advantages of our Radiotheranostic-based approach include:
Our Radiotheranostic solution is an antibody-based platform that can be easily equipped with either a diagnostic or therapeutic radionuclide. This allows for precise patient selection, as the diagnostic version identifies real-time, whole-body presence of the target of treatment, while the same antibody now armed with a therapeutic radionuclide delivers a high absorbed dose for local treatment.
It is a precise, targeted, and effective radiopharmaceutical with improved tumor uptake and reduced adverse effects.
It holds great potential to be a superior therapeutic option for patients who do not respond to current ADC treatment or other later-line systemic therapies.
OUR TECHNOLOGY
A single monoclonal antibody platform, elegantly engineered using click‑chemistry technology, enables precise diagnostic and therapeutic applications. The antibody can be labeled with a Positron Emission Tomography (PET) imaging radionuclide for diagnosis or with beta or alpha‑emitting radionuclides for targeted therapy. It selectively binds to Trop-2-expressing tumor cells while sparing healthy, non-expressing tissues
Diagnostic (Selection & Dosing): Imaging confirms target‑positive patients and predicts therapeutic response.
Therapy: Same antibody delivers precise, target‑matched treatment via ionizing radiation.
Follow‑Up: Repeat imaging tracks target changes and treatment responses.
Trophoblast cell-surface antigen 2 (TROP-2) is a transmembrane glycoprotein frequently overexpressed across many solid tumors with a low/absent expression in healthy tissues, making it a true pan‑cancer therapeutic target with broad applicability and a large potential patient population.
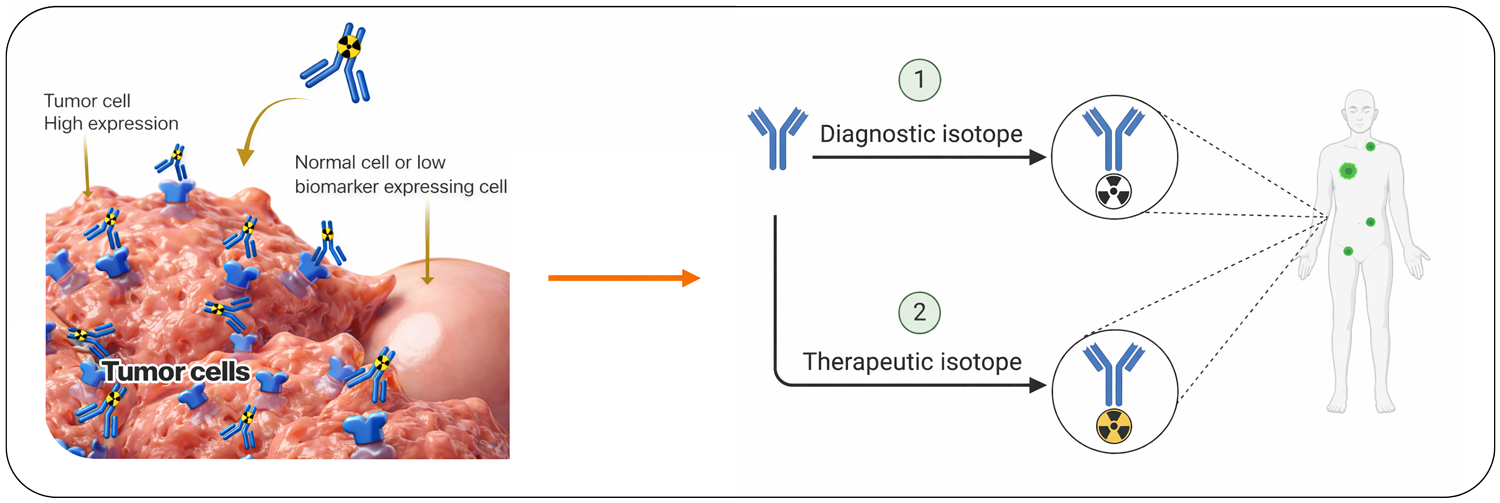
An innovation based on a novel, targeted all-in-one, radiolabeled TROP-2 monoclonal antibody platform, elegantly designed through site-specific click chemistry technology. Diagnostic isotopes such as 89Zr are suitable for antibody imaging with PET, while for example radionuclides of Lu-177 (beta emitter), Tb-161 (beta emitter with Auger Meitner electrons) or Ac-225 (alpha emitter) are well-suitable for molecular radiotherapy.
OUR TEAM
Our mission statement
To advance cancer treatment by developing radiotheranostic tools that enable more precise diagnosis and targeted therapy to those who need it most

Klas Bratteby
Ph.D., CEO,
Co-founder
Thuy Tran
Ph.D., CSO,
Co-founder
Associate Professor at Karolinska Institutet and Senior Consultant Medical Oncologist at Docrates Cancer Center. Principal investigator for multiple clinical studies in oncology and experienced academic leader. Recognized key opinion leader in precision oncology and nuclear medicine applications.
Jonathan Siikanen
Ph.D., CTO,
Co-founder
Senior Research Specialist at Karolinska Institutet and Specialist in Medical Physics at the Theranostics Trial Center, Karolinska University Hospital. Internationally recognized expert in radionuclide production with extensive experience in radiation safety and nuclear medicine technologies.

Karl
Petterson Palm,
M.D. & Ph.D.-candidate, COO, Co-founder


Associate Professor and research group leader at Karolinska Institutet with more than 15 years of leadership in radiopharmaceutical R&D. Co-founder and head of the Theranostics Trial Center at Karolinska University Hospital. Has led more than 10 radiopharmaceutical assets into Phase I clinical trials and is co-inventor of radiotherapeutic antibodies licensed to major pharmaceutical companies.
Researcher with over 8 years of experience in radiopharmaceutical R&D at Karolinska Institutet and the Theranostics Trial Center. Has contributed to the translation of multiple radiopharmaceutical assets from preclinical development to first-in-human studies. Co-inventor of intellectual property licensed to Tetrakit.


Renske Altena
Ph.D., CMO,
Co-founder
PhD-student and researcher trained at Karolinska Institutet with extensive expertise in radiopharmaceutical development. Experienced in both in vitro and in vivo studies supporting the discovery and preclinical evaluation of novel radiopharmaceutical candidates.
